usp endotoxin limits|Iba pa : Tagatay For intrathecally administered radiopharmaceuticals, the endotoxin . https://baraha88.com?code=1396627 Ito po LEGIT meron yarn free coins Try nyo guys kapag nanalo kayo ng 100 pesos pd nyo agad cash out at lahat ng uri ng.
usp endotoxin limits,USP's revision of the standard for Bacterial Endotoxins has been approved by the Pharmacopeial Discussion Group (PDG) and will be incorporated with the second supplement within USP 35- NF 30.
For intrathecally administered radiopharmaceuticals, the endotoxin .
The endotoxin limit for parenteral drugs, defined on the basis of dose, is equal to K/M, 4 where K is the threshold human pyrogenic dose of endotoxin per kg of body weight, and .
Iba paFor intrathecally administered radiopharmaceuticals, the endotoxin limit toxin Unit per mL. is obtained by the formula 14 EU/ V . For formulations (usually anticancer

The Center for Devices and Radiological Health (CDRH) has adopted the USP Endotoxin Reference Standard and limits for medical device extracts expressed in EU/mL.usp endotoxin limits Iba paThe Center for Devices and Radiological Health (CDRH) has adopted the USP Endotoxin Reference Standard and limits for medical device extracts expressed in EU/mL.Water for Injection, Sterile Water for Injection and Sterile Water for Irrigation have an allowable endotoxin limit of 0.25 Endotoxin Units (EU)/ml. (EU=Unit of measurement for .
The lower the dose, the higher the limit per unit dose. Think about it this way: If the dose is 1 mg/kg/hr, the endotoxin limit is (5 EU/kg/hr) ÷ (1 mg/kg/hr) = 5 EU/mg; If the dose is 10 mg/kg/hr, the .Endotoxin limit: the endotoxin limit for active substances administered parenterally, defined on the basis of dose, is equal to: = threshold pyrogenic dose of endotoxin per .Description of scope and application: This proposed general information chapter will provide guidelines on Bacterial Endotoxins Test relative to but not limited to: control standard .
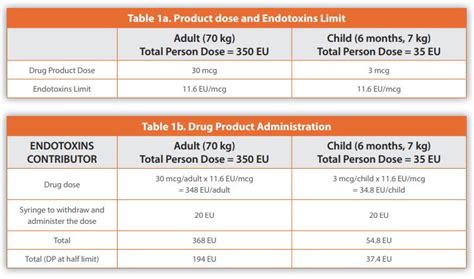
Endotoxin Limit The endotoxin limit for medicinal products administered parenterally, is defined on the basis of dose. Calculate the endotoxin limit as follows: Result = K /M K .

The purpose of this information chapter is to provide additional background information and guidance for the performance and proper application of the compendial bacterial .
44 endotoxin limits for that investigational drug at a time when the manufacturing process for that . 117 parenteral drugs recommended in USP General Chapter <85> Bacterial Endotoxins Test. areThe <85> Bacterial Endotoxins Test General Chapter was incorporated into and became official with the Second Supplement to USP 35–NF 30. Should you have any questions about this General Chapter, please contact Rahdakrishna Tirumalai (301-816-8339 or [email protected] ). For any questions about the PDG and its processes, please see the .Endotoxin limit = K /M = 100 EU/ m2 / 30 mg/m2 = 3.33 EU/mg Medical Devices USP chapter <161>4 set generic endotoxin limits of 20 EU/device for most devices labeled as non-pyrogenic and 2.15 EU/device for devices that contact the cerebrospinal fluid (CSF). As an aqueous solution is required for the BET,After the withdrawal of FDA’s 1987 guideline on the LAL test, there were gaps in the regulatory documents regarding certain aspects of endotoxin testing like RSE/CSE standardization. On 01 December 2019, USP therefore published General Chapter <1085> Guidelines on the Endotoxins Test. This new chapter aims to “provide additional .Endotoxin Unit (EU) is a unit of biological activity of the USP Reference Endotoxin Standard. Summary This document describes a uniquely simple system for assuring that compounded sterile products are free of endotoxin (pyrogen) within limits set by the Pharmacopeia (USP) or consistent with current scientific opinion.usp endotoxin limitsThe newest United States Pharmacopeia (USP) Chapter <1085> Guidelines on Endotoxins Testing fills in the regulatory gaps that have resulted following the withdrawal of the 1987 FDA Guideline and implementation of its replacement Q&A. Below are answers to some of the most frequently asked questions pertaining to USP Chapter <1085>.The endotoxin units set by United States Pharmacopoeia (USP), and the techniques specified by USP for endotoxin testing are described. Endotoxin limits for preclinical research animal models were derived based on the threshold pyrogenic human dose of 5 E.U. per kg. The limits calculated would act as a guideline for endotoxin limits in .
http://w w w.usppf.com /pf/pub/i ndex.htm l 1/1
usp endotoxin limits|Iba pa
PH0 · usp endotoxin reference standard
PH1 · usp 161 endotoxin
PH2 · how to calculate endotoxin limit
PH3 · fda guidance pyrogen and endotoxin testing
PH4 · fda endotoxin guidance
PH5 · endotoxin limits for parenteral drugs
PH6 · endotoxin limit for injectable products
PH7 · bacterial endotoxin test procedure
PH8 · Iba pa